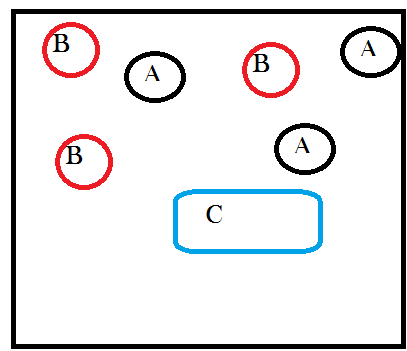
Next let’s give A and B the possibility, if they collide in just the right way, of joining together to form a new molecule that is a combination of A and B. We’ll call this new molecule “C” and we can write out the chemical reaction as:
A + B → C
A complicated reaction, I know. Ok so “ignoring energetic considerations” means assuming that the temperature and pressure in our container don’t change as a result of A and B combining to make C. This is an artificial assumption that I’m introducing so that we can analyze only the effects of matter transformations. Note that it takes two molecules, one each of A and B, to make one C. This means that every time this happens there is one less particle in our system because two things became one thing. But also the first time it happens there is one new particle in our system, a C exists where there were no C’s before. So after one reaction, our system will look like this:
See how I took away one A and oneB and replaced them with a new thing called C? Still with me?